Protein Engineering
Improving the solubility and stability of biologics
The proteostatic quality control machinery keeps protein aggregation in check within cells. When proteins are produced for research, therapeutic or industrial applications, however, they encounter artificial conditions without the help available within cells. This often leads to aggregation which is the primary factor impeding the production and application of many biologicals like monoclonal antibodies and enzymes.
The Switch laboratory has developed a method to adapt the primary sequence of proteins to make them more resistant to aggregation, while maintaining their stability and function. Our rational protein design strategy, called the Solubis method [1,2], detects the linear segments that drive aggregation within a protein sequence and predicts mutations that reduce the aggregation propensity of these segments without affecting the thermodynamic stability of the protein. Reducing the aggregation propensity often leads to proteins with increased solubility and higher production yields.
SolubiS uses the TANGO algorithm [3] to predict aggregation prone regions (APRs) in the primary sequence. Then it uses the FoldX empirical all-atom force-field [4,5] to calculate the contribution of each APR to the local stability of the protein. Force fields are in silico simulations of the physical rules of the material world and FoldX helps to identify APRs that are most likely to occur in aggregation-competent conformations.
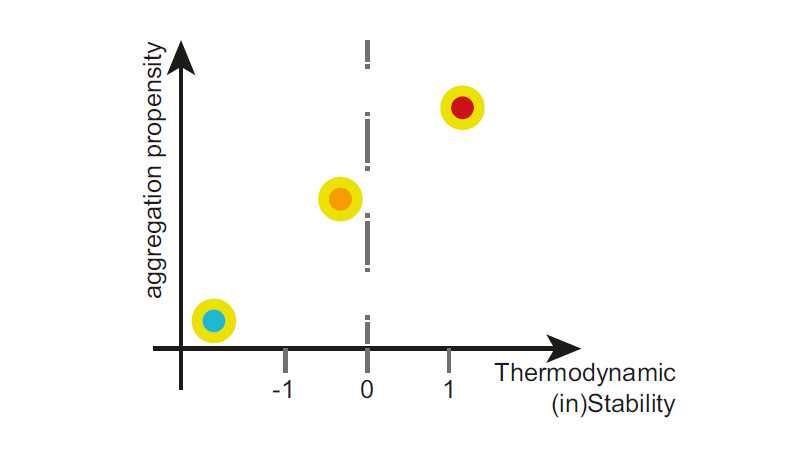
We display the total aggregation propensity of a protein using so-called stretch plots (see below), where each APR is represented as a single point in a two-dimensional space with the aggregation propensity is on the y-axis and its contribution to protein stability is on the x-axis. SolubiS focuses on unstable and highly aggregation prone ‘critical’ APRs that are most likely to drive aggregation (red dot in the plot below).
SolubiS then systematically scans the identified aggregation regions for mutations that could reduce protein aggregation, while maintaining the stability and structure of the protein. Often a combination of mutations is required to maximally suppress aggregation, thereby targeting multiple weak points in the protein. We have validated SolubiS in several case studies [1,6-9].
For example, we have used it to improve the solubility of an important component of recombinant Anthrax vaccines. Immune response against the so-called Protective Antigen (PA) confers protection against Bacillus anthracis infection, but the recombinant PA is aggregation-prone in aqueous buffers which limits its production and storage. We have produced a PA variant that has improved thermodynamic stability and increased aggregation resistance at 40 °C.
The red dot in the stretch-plot indicates the “critical” APR in wild type Protective Antigen and the small red circle shows its location within the crystal structure (PDB ID: 1T6B). The large red circle shows the same area of the aggregation resistant mutant (T576E/S559 L), based on a crystal structure (PDB ID: 5FR3). Mutations are highlighted. Images of protein structures were generated using YASARA Structure. From [1]
We have also used SolubiS to reduce the aggregation propensity of monoclonal antibodies without affecting their desired function in collaboration with Boehringer Ingelheim [8]. The SolubiS method is patent protected both in Europe and in the United States.
References
- van der Kant R, van Durme J, Rousseau F, Schymkowitz J (2019) SolubiS: Optimizing Protein Solubility by Minimal Point Mutations. Methods Mol Biol 1873:317-333. doi:10.1007/978-1-4939-8820-4_21
- Van Durme J, De Baets G, Van Der Kant R, Ramakers M, Ganesan A, Wilkinson H, Gallardo R, Rousseau F, Schymkowitz J (2016) Solubis: a webserver to reduce protein aggregation through mutation. Protein engineering, design & selection : PEDS 29 (8):285-289. doi:10.1093/protein/gzw019
- Fernandez-Escamilla AM, Rousseau F, Schymkowitz J, Serrano L (2004) Prediction of sequence-dependent and mutational effects on the aggregation of peptides and proteins. NATURE BIOTECHNOLOGY 22 (10):1302-1306. doi:10.1038/nbt1012
- Schymkowitz JW, Rousseau F, Martins IC, Ferkinghoff-Borg J, Stricher F, Serrano L (2005) Prediction of water and metal binding sites and their affinities by using the Fold-X force field. Proc Natl Acad Sci U S A 102 (29):10147-10152. doi:10.1073/pnas.0501980102
- Guerois R, Nielsen JE, Serrano L (2002) Predicting changes in the stability of proteins and protein complexes: A study of more than 1000 mutations. Journal of Molecular Biology 320 (2):369-387. doi:10.1016/S0022-2836(02)00442-4
- Siekierska A, De Baets G, Reumers J, Gallardo R, Rudyak S, Broersen K, Couceiro J, Van Durme J, Schymkowitz J, Rousseau F (2012) alpha-Galactosidase aggregation is a determinant of pharmacological chaperone efficacy on Fabry disease mutants. J Biol Chem 287 (34):28386-28397. doi:10.1074/jbc.M112.351056
- Ganesan A, Siekierska A, Beerten J, Brams M, Van Durme J, De Baets G, Van der Kant R, Gallardo R, Ramakers M, Langenberg T, Wilkinson H, De Smet F, Ulens C, Rousseau F, Schymkowitz J (2016) Structural hot spots for the solubility of globular proteins. Nat Commun 7:10816. doi:10.1038/ncomms10816
- van der Kant R, Karow-Zwick AR, Van Durme J, Blech M, Gallardo R, Seeliger D, Assfalg K, Baatsen P, Compernolle G, Gils A, Studts JM, Schulz P, Garidel P, Schymkowitz J, Rousseau F (2017) Prediction and Reduction of the Aggregation of Monoclonal Antibodies. J Mol Biol 429 (8):1244-1261. doi:10.1016/j.jmb.2017.03.014
- van der Kant R, Bauer J, Karow-Zwick AR, Kube S, Garidel P, Blech M, Rousseau F, Schymkowitz J (2019) Adaption of human antibody lambda and kappa light chain architectures to CDR repertoires. Protein engineering, design & selection : PEDS 32 (3):109-127. doi:10.1093/protein/gzz012